Products
Features
- Save time — just add to cells and incubate
- Efficient transfection with reduced effort
- Consistency across multiple experiments
Product Details
FlexiTube siRNA Premix is an optimized mixture of siRNA and transfection reagent. Premixing of siRNA and reagent saves time by eliminating mixing and complex formation protocol steps. In addition, use of FlexiTube siRNA Premix removes the effort involved in estimating optimal siRNA-reagent ratios. FlexiTube siRNA Premix is available for human and mouse genes.
Performance
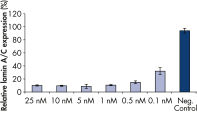
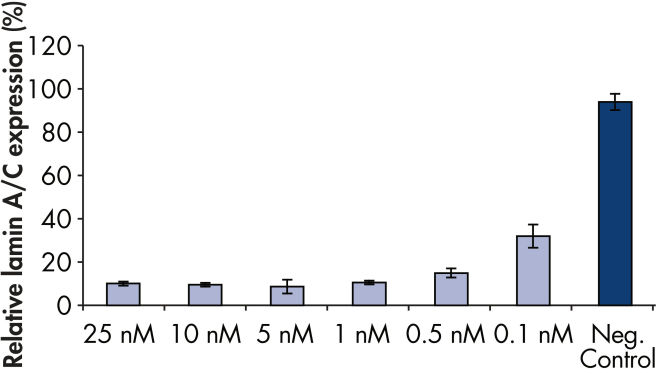
The ratio of reagent to siRNA in FlexiTube siRNA Premix provides highly efficient transfection and gene knockdown (see figures " Rapid, efficient knockdown" and " Phenotype analysis after knockdown").
FlexiTube siRNA Premix contains FlexiTube siRNA, which is designed using highly innovative and sophisticated HP OnGuard siRNA Design to deliver high potency and specificity. siRNAs are designed using neural-network technology based on a large data set from RNAi experiments. siRNA design is then checked for homology to all other sequences of the genome using an up-to-date, nonredundant sequence database and a proprietary homology analysis tool that incorporates exciting design features.
Performance guarantee
FlexiTube siRNA Premixes come with a one-time-only replacement offer. If several FlexiTube siRNA Premixes for the same target gene are ordered and at least 2 of them do not provide ≥70% target gene knockdown, QIAGEN will provide 2 additional siRNAs free of charge, once only. You will be asked to provide supporting data, demonstrating that the siRNA failed to knock down the target gene by at least 70% at the mRNA level under appropriate transfection conditions. Supporting data should include transfection efficiency data, quantitative silencing data, and data showing ≥70% knockdown of a positive control. This offer is valid for up to 6 months after the date of delivery.
Principle
Procedure
FlexiTube siRNA Premix is ready-to-use for cell transfection. Simply add to cells and incubate. This is a significant time and labor saving compared with a typical transfection experiment in which siRNA is diluted, mixed with transfection reagent, and then the mixture is incubated to allow siRNA-reagent complex formation prior to adding the complexes to the cells and incubating.
With FlexiTube siRNA Premix, RNAi experiments get off to a faster start, as there is no need to optimize siRNA to reagent ratio. Tedious optimization experiments involving multiple transfections are minimized or eliminated, as FlexiTube siRNA Premix provides siRNA and reagent premixed at an optimal ratio.
Multiple transfections can be performed from a single FlexiTube siRNA Premix. This reduces variability, enabling consistency across experiments and more reliable results.
Applications
FlexiTube siRNA Premix is ideal for functional genomics or pathway analysis with small numbers of gene targets.
Supporting data and figures
Rapid, efficient knockdown.
Specifications
| Features | Specifications |
|---|---|
| Design | HP OnGuard design |
| Target sequence provided | Yes |
| Modification | No |
| siRNA per target gene | variable |
| Format | Tube |
| Number of transfections | 50 standard transfections in 24-well format |
| Guarantee/validation | for validated siRNAs |
| Species | Human, mouse |
| Scale or yield | 0.75 nmol |