Proteinase K - Powder (100 mg)
Cat no. / ID. RP100B
Features
- High activity in elevated temperatures (up to 56°C) and under denaturing conditions
- Stable over a wide pH range of 4.0–12.5 for use in a variety of applications
- Exceptional purity with a decreased amount of host DNA recombinant enzyme for no cross-reactivity contamination in NGS/diagnostics
Product Details
Proteinase K is a subtilisin-related serine protease derived from the Parengyodontium album (Tritirachium album) that is active under a wide range of reaction conditions, including elevated temperatures and the presence of Sodium Dodecyl Sulfate (SDS).
It is a broad-spectrum endopeptidase with a very high specific activity allowing highly effective digestion of proteins, including DNases and RNases, during nucleic acid preparations without compromising the integrity of isolated DNA or RNA.
The enzyme is expressed in Pichia pastoris, and undergoes extensive purification to yield the highest quality product. An additional purification technology significantly increases solubility (2.5 fold), increases specific activity, and produces remarkable purity with DNA content ≤0.1 pg/mg for Proteinase K Ultrapure.
Proteinase K is free of exonucleases, endonucleases, and ribonucleases.
Proteinase K Ultrapure is developed with additional purification technology resulting in its significantly increased solubility (2.5 fold), increased specific activity, and remarkable purity with DNA content ≤0.1 pg/mg.
One unit of Proteinase K hydrolyzes urea-denatured hemoglobin producing the color equivalent of 1 μmol tyrosine per 1 minute at 37°C and pH 7.5 (Folin & Ciocalteu’s method), 1 U = 1 mAnsonU.
Performance
| Assay | Proteinase K Powder and Solution Specification | Proteinase K Ultrapure Specification |
|---|---|---|
| Protein content | >70% | >70% |
| Solubility in water | ≥20 mg/mL | ≥50 mg/mL |
| Activity | ≥30 U/mg lyophilizate ≥40 U/mg protein ≥800 U/mL liquid |
≥35 U/mg lyophilizate ≥45 U/mg protein |
| DNA content | ≤10 pg/mg ≤200 pg/mL |
≤0.1 pg/mg |
| Single-stranded exonuclease | Not detected | Not detected |
| Double-stranded exonuclease | Not detected | Not detected |
| Double-stranded endonuclease | Not detected | Not detected |
Principle
Proteinase K is a recombinant Parengyodontium album (Tritirachium album) 28.9 kDa broad-spectrum serine protease expressed in Komagataella phaffii (Pichia pastoris) used for protein digestion and contamination removal during nucleic acid preparation. It cleaves the peptide bond adjacent to the carboxyl group of aliphatic, aromatic, and other hydrophobic amino acids. Because of its broad specificity and high stability at a wide range of pH and temperature, Proteinase K is routinely used in numerous molecular biology applications such as the isolation of genomic, plasmid, and high molecular weight DNA, RNA, and in inactivation of RNases and DNases. It is also used for effective digestion of structural proteins, chromatins, and inactivation of nucleases and RNases.
High-quality sample preparation is an essential step before the actual molecular analysis of the genetic material. This procedure includes multiple stages such as sample isolation, purification, and concentration of the target product, where Proteinase K plays an important role. Despite various reagents used in the process, the initial focus should be on Proteinase K, which certified quality assures high purity of tested nucleic acids.
Proteinase K powder
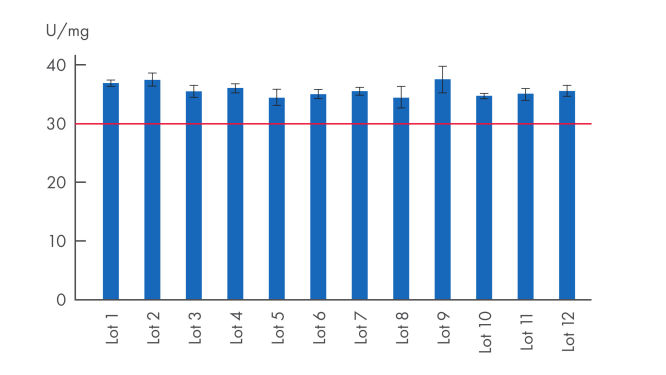
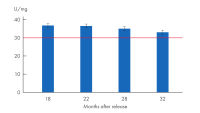
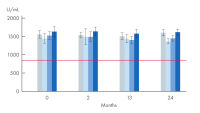
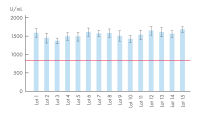
Proteinase K powder is a lyophilized, white powder, highly active with exceptional lot-to-lot consistency (see figure “ High lot-to-lot consistency Proteinase K powder”), ensuring reproducibility for stable working conditions, repeatable and reliable experiment results. Proteinase K powder is highly stable when stored at -20°C (see figure “ Proteinase K powder stability”).
Proteinase K powder specifications:
- No detected exonuclease, endonuclease or RNase activity
- Solubility in water ≥20 mg/mL; activity ≥30 U/mg lyophilizate; specific activity ≥40 U/mg protein; protein content ≥70%; DNA content ≤10 pg/mg
- Shipping conditions: Ambient temperature
Proteinase K solution
Proteinase K solution is a clear, colorless liquid with long-term stability when stored at room temperature. No need to refrigerate (see figure „ High stability Proteinase K liquid”). Established manufacturing process ensures high lot-to-lot consistency (see figure “ High lot-to-lot consistency Proteinase K liquid”).
Proteinase K solution specifications:
- No detected exonuclease, endonuclease or RNase activity
- Concentration ≥20 mg/mL; activity ≥ 800 U/mL; DNA content ≤ 200 pg/mL; bioburden ≤1 CFU/mL
- Shipping conditions: Ambient temperature
- Storage buffer: 10 mM Tris-HCl, pH 7.5; 1 mM (CH3COO)2Ca; 50% glycerol
Proteinase K Ultrapure
Proteinase K Ultrapure is a white, lyophilized powder with increased activity and solubility for most demanding applications and decreased DNA content for sensitive applications and no cross-contamination.
Proteinase K Ultrapure specification:
- No detected exonuclease, endonuclease or RNase activity
- Solubility in water ≥ 50 mg/mL; activity ≥ 35 U/mg lyophilizate; specific activity ≥45 U/mg protein; protein content ≥70%
- DNA content ≤0.1 pg/mg
- Shipping conditions: Ambient temperature
Procedure
Quality Control
Protein concentration was determined by measuring absorbance at 280 nm.
The presence of exonuclease was determined by gel electrophoresis of 1 μg of HindIII-digested λ DNA with 50 μg of Proteinase K incubated for 16 hours at 37°C.
The presence of endonuclease was determined by gel electrophoresis of 1 μg pUC19 DNA with 40 μg of Proteinase K incubated for 16 hours at 37°C.
The presence of RNase activity was determined by gel electrophoresis of 2 µg rRNA from E. coli with 20 μg of Proteinase K incubated for 4 hours at 37°C.
Usage
Proteinase K stock solution preparation
20 mg/mL solutions: use purified water for immediate use.
20–50 mg/mL solutions: use 50 mM Tris-HCl, pH = 7.5-8.0, 1-5 mM Ca2+ (calcium chloride, calcium acetate) for immediate use; or 10 mM Tris-HCl, pH = 7.5-8.0, 1-5 mM Ca2+ (calcium chloride, calcium acetate), 50% glycerol for long-term storage.
Applications
This product is available for molecular biology applications such as:
- Extracting DNA and RNA from different starting materials
- Eliminating DNases and RNases during nucleic acid isolation
- Purifying samples contaminated with different protein
- Automating isolation stations
Supporting data and figures
High lot-to-lot consistency Proteinase K powder
Consistent quality from lot-to-lot – lyophilized Proteinase K. In internal comparison tests, Proteinase K shows consistently high levels of activity with limited deviation. The red line represents the declared minimum activity of lyophilized proteinase K (≥30 U/mg).