✓ 24/7 automatic processing of online orders
✓ Knowledgeable and professional Product & Technical Support
✓ Fast and reliable (re)-ordering
Cat. No. / ID: 51704
✓ 24/7 automatic processing of online orders
✓ Knowledgeable and professional Product & Technical Support
✓ Fast and reliable (re)-ordering
Features
- Isolation of bacterial DNA from swabs and body fluids
- Effective depletion of host DNA
- Optimized mechanical and chemical cell lysis
- Ultra-clean columns to minimize contamination risk
Product Details
The QIAamp DNA Microbiome Kit is a dedicated solution for the purification and enrichment of bacterial microbiome DNA from swabs and body fluids. Effective depletion of host DNA during the purification process maximizes bacterial DNA coverage in next generation sequencing analysis and allows highly sensitive 16S rDNA-based microbiome analysis and whole metagenome shotgun sequencing studies.
Performance
Swabs and body fluids contain DNA from host human or animal cells as well as microbial cells. In metagenomic studies, total DNA is standardly isolated and analyzed, however, host human or animal DNA greatly outweighs the microbial DNA, which can hamper microbiome analysis. In fact, a key finding of the whole metagenome shotgun sequencing of different sample types conducted by the Human Microbiome Project was that up to 99% of the sequencing reads corresponded to the human genome, so that at most 1% were of microbial nature. In contrast to 16S rDNA sequencing, whole metagenome shotgun sequencing can add valuable insight to microbiome studies, such as the presence of virulence factors, antibiotic resistance, or metabolic networks. Thus, maximized coverage of microbial reads greatly enhances analysis power.
Removal of host DNA increases the coverage of microbial reads in sequencing experiments. The QIAamp DNA Microbiome Kit efficiently removes host DNA by differential lysis of host cells and subsequent enzymatic digestion of host DNA. Then, using a combination of mechanical and chemical lysis, intact cells are efficiently lysed and the released bacterial DNA is purified using proven QIAamp chemistry and decontaminated QIAamp UCP spin columns (see figure Purification procedure with integrated host DNA removal).
Enhanced resolution of bacterial DNA in whole metagenome shotgun sequencing experiments
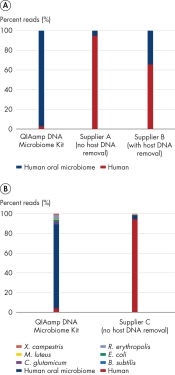
The high percentage of reads attributed to the host genome in whole metagenome shotgun sequencing experiments makes proper assembly of microbial datasets time consuming and resource intensive, even with sophisticated bioinformatics tools. By removing host DNA in a sample, the QIAamp DNA Microbiome Kit provides samples with enriched bacterial composition for whole metagenome shotgun sequencing. A comparison of reads from whole metagenome shotgun sequencing experiments was performed on DNA isolated from human buccal swabs, where samples were prepared with either the QIAamp DNA Microbiome Kit or solutions from 2 other suppliers. Sequencing of samples prepared with the QIAamp DNA Microbiome Kit resulted in less than 5% human reads, greatly reduced compared to over 90% reads with a kit that does not include host DNA removal and 35% with a kit that does. In a second experiment, samples were spiked with known cultured bacteria. Bacterial DNA were recovered with higher representation compared to a solution without host DNA removal (see figure Effective host DNA removal enhances whole metagenome shotgun sequencing results).
Improved amplification for 16S rDNA sequencing
16S rDNA sequencing is commonly used to determine relative microbial community composition. The QIAamp DNA Microbiome Kit enables the most efficient amplification of the V4 region of DNA purified from buccal swabs when compared to 3 other purification kits (see figure Efficient amplification and sequencing of 16S rDNA). Sequencing results of the amplified V4 region from 2 samples purified with the QIAamp DNA Microbiome Kit also exhibit the expected bacterial composition representative of a human oral microbiome.
Optimized cell lysis to minimize sample preparation biasDifferences in cell wall morphology render microbes differentially susceptible to different lysis methods. For example, bacteria with thick cell walls, rich in lipids and polysaccharides, tend to be underrepresented in samples prepared via enzymatic lysis. The result is that any one lysis method can introduce a bias in the representation and relative composition of a microbiome. The QIAamp DNA Microbiome Kit uses a combination of chemical and mechanical lysis, optimized to minimize bias introduced by sample preparation. Furthermore, the Ultra Clean Production (UCP) spin columns provided in the QIAamp DNA Microbiome Kit undergo a proprietary cleaning process to minimize the risk of contamination.
To examine sample preparation bias, a model microbiome with known numbers of colony forming units (CFUs) of 6 different bacteria was created from cultures. Samples of the model microbiome were prepared for sequencing using the QIAamp DNA Microbiome Kit or alternative kits. The samples processed with the QIAamp DNA Microbiome Kit exhibited the best overall representation of the model microbiome composition compared to all other methods used (see figure Minimal sample preparation bias).
See figures
Principle
Procedure
See figures
Applications
The QIAamp DNA Microbiome Kit purifies and enriches bacterial microbiome DNA from mixed samples for use in sensitive downstream applications such as:
- Whole genome sequencing analysis
- Highly-sensitive 16S rDNA-based microbiome analysis
- Metagenomic shotgun sequending studies
Supporting data and figures
Effective host DNA removal enhances whole metagenome shotgun sequencing results.