✓ 全天候自动处理在线订单
✓ 博学专业的产品和技术支持
✓ 快速可靠的(再)订购
pQE-TriSystem Vector
Cat. No. / ID: 33903
25 µg pQE-TriSystem Vector DNA
登录 要查看您的账户定价。
pQE-TriSystem Vector 旨在用于分子生物学应用。该产品不能用于疾病诊断、预防和治疗。
✓ 全天候自动处理在线订单
✓ 博学专业的产品和技术支持
✓ 快速可靠的(再)订购
特点
- 无需耗时的亚克隆程序
- 在昆虫或哺乳动物细胞中实现翻译后修饰
- 一个构建体在三个表达系统中提供高效表达
产品详情
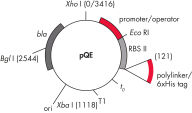
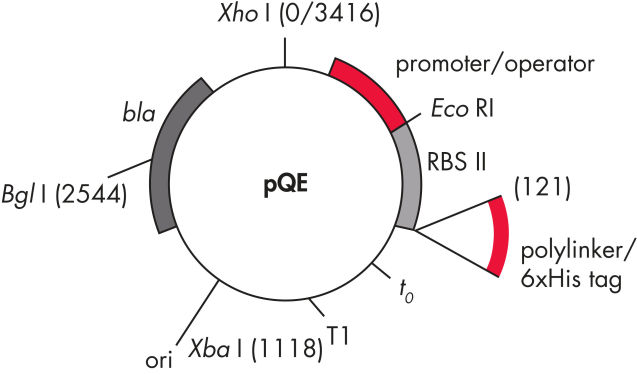
pQE TriSystem Vector 允许利用单一载体在三个不同表达系统中进行 His 标签蛋白的高水平表达。T5 启动子/lac 操作子转录–翻译系统用于在 E. coli 中进行表达,p10 启动子用于在昆虫细胞中进行基于杆状病毒的表达,CAG(CMV/肌动蛋白/珠蛋白)启动子用于在哺乳动物细胞中进行表达。
绩效
pQE-TriSystem 载体包含 CAG、T5 和 p10 启动子,分别支持在哺乳动物、E. coli 和杆状病毒感染的昆虫细胞中进行 6xHis 标签蛋白表达(参见图片 pQE TriSystem)。可以使用强 T5 启动子在细菌表达系统中进行初步研究,该启动子可被 E. coli 聚合酶识别,并允许在任何 E. coli 菌株中高效表达蛋白质。如果需要在哺乳动物或昆虫细胞中表达——例如,为了实现翻译后修饰——可以使用相同的构建体,而不需要耗时的亚克隆程序。
查看图表
原理
QIAexpress pQE 载体将强效的噬菌体 T5 启动子(可被 E. coli RNA 聚合酶识别)与双 lac 操作子抑制模块相结合,以在 E. coli 中提供严格调控的高水平重组蛋白表达。在存在高水平 lac 阻遏物的情况下,蛋白质合成被有效阻断,并且细胞毒性构建体的稳定性得以增强。pQE 载体(参见表格和图片 pQE 载体)能够将 6xHis 标签置入重组蛋白的 N-末端或 C-末端。
| 元件 | 说明 |
| 1.优化的启动子/操作子元件 |
由噬菌体 T5 启动子和两个 lac 操作子序列组成, 这增加了 lac 阻遏物结合的可能性,并确保 强效 T5 启动子的有效抑制 |
| 2.合成核糖体结合位点 RBSII | 用于高效翻译 |
| 3.His 标签编码序列 | 5' 或 3' 到多位点人工接头克隆区 |
| 4.翻译终止密码子 | 在所有阅读框架中,用于方便地制备表达构建体 |
| 5.两个强转录终止子 |
t0 来自噬菌体 λ,T1 来自 E. coli 的 rrnB 操纵子,以阻止 连读转录并确保表达构建体的稳定性 |
|
6.ColE1 复制起点 |
来自 pBR322 |
| 7. β-内酰胺酶基因 (bla) | 赋予氨苄青霉素耐药性 |
查看图表
程序
将编码目标蛋白质的插入片段克隆到合适的构建体中,并转化至合适的 E. coli 菌株中进行表达。通过添加 IPTG 来诱导表达。载体 pQE TriSystem 构建体可以转化至 E. coli 中,用作昆虫细胞中重组蛋白表达的穿梭载体,或转染至哺乳动物细胞中。
应用
QIAexpress Expression System 提供适用于许多应用的蛋白质高水平表达,包括:
- 功能性构象活性蛋白质的纯化
- 在变性条件下进行纯化用于抗体生产
- 结晶用于三维结构测定
- 涉及蛋白质-蛋白质和蛋白质-DNA 相互作用的检测
辅助数据和图表
pQE 载体。
表中列出的编号元素。
Specifications
| Features | Specifications |
|---|---|
| In-frame cloning necessary | 是 |
| Expression | 体内 |
| Tag removal sequence | 否 |
| Expression species | E.coli,哺乳动物和昆虫细胞 |
| Tag | 6xHis 标签 |
| N- or C-terminal tag | C-末端标签 |
| All three reading frames provided | 否 |
资源
载体序列 (7)
安全数据表 (1)
产品选择指南 (1)
试剂盒操作手册 (1)
Safety Data Sheets (1)
Certificates of Analysis (1)
FAQ
Is it possible to use QIAGEN's pQE-TriSystem Vectors with the EasyXpress Protein Synthesis Insect Kit?
How can I increase expression of my 6xHis-tagged protein in E. coli?
How can I increase the amount of soluble recombinant protein in E. coli expression?
What is the origin of replication and the plasmid copy number of the pQE vectors?
How should I propagate pQE expression plasmids?
Do pQE vectors contain the CAT gene?
How can I be sure that I am harvesting my induced bacterial culture at the best time point for protein expression?