✓ 24/7 automatic processing of online orders
✓ Knowledgeable and professional Product & Technical Support
✓ Fast and reliable (re)-ordering
Cat. No. / ID: 74171
✓ 24/7 automatic processing of online orders
✓ Knowledgeable and professional Product & Technical Support
✓ Fast and reliable (re)-ordering
Features
- Simple and reliable automated processing for time and cost savings
- Flexible procedures for isolating total RNA or total RNA, including miRNA
- Protocols for various starting materials and desired size distribution
- High-quality purified RNA ready for use in any downstream application
- Consistent RNA yields from small amounts of starting material
Product Details
The RNeasy 96 QIAcube HT Kit enables simple, automated purification of total RNA or total RNA, including miRNA, from animal and human cells and tissue samples on the QIAcube HT system. Using proven RNeasy silica-membrane technology in a convenient 96-well format, contaminants and inhibitors are removed to yield high-quality RNA ready for downstream analysis.
Performance
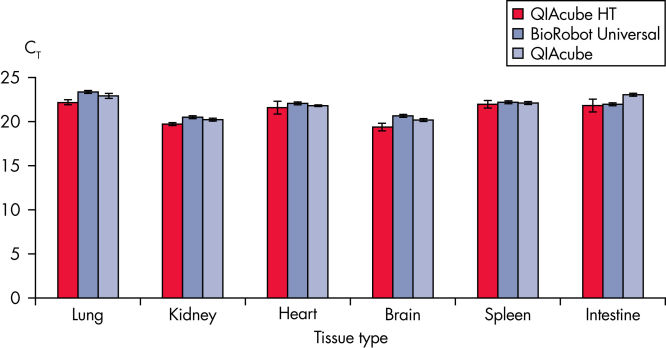
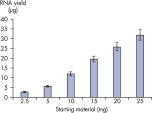
This economical, easy-to-use system provides the same results as other trusted QIAGEN purification technologies (see figure Automated purification of RNA from tissues). Analysis of QIAcube HT purified samples shows high RNA Integrity Scores (RIS) when analyzed with the QIAxcel Advanced system, and performance similar to other automated QIAGEN purification solutions (see figure High-quality RNA from tissues).
The RNeasy 96 QIAcube HT Kit provides consistent yields from small amounts of starting sample material (see table Typical total RNA yields from cells with the RNeasy 96 QIAcube HT Kit).
| Cell line | Source | RNA yield (µg per 105 cells)* |
|---|---|---|
| HeLa | Human cervical carcinoma | 1.6 |
| LMH | Chicken hepatoma | 1.3 |
| COS-7 | Monkey kidney, SV-40 transformed | 3.1 |
| Huh7 | Human hepatoma | 2.0 |
| Jurkat | Human T-cell leukemia | 1.4 |
| K-562 | Human chronic myelogenous leukemia in blast crisis | 1.9 |
See figures
Principle
The RNeasy 96 QIAcube HT Kit enables automated purification of total RNA or total RNA, including miRNA, from 5 x 105 animal or human cells or <40 mg tissue using the QIAcube HT instrument. Six instrument protocols are available, depending on the starting material and desired size distribution of purified RNA. Cell samples can be purified directly using the RNeasy 96 QIAcube HT Kit and the dedicated kit protocol. Tissue samples (<40 mg) and samples for purification of small RNA are processed following sample lysis in QIAzol Lysis Reagent. The procedures yield high-quality RNA that performs well in downstream applications.
The RNeasy 96 QIAcube HT Kit combines the selective binding properties of a silica-based membrane with a high-throughput 96-well format, and is designed for fully-automated, simultaneous processing of 24–96 samples on the QIAcube HT instrument.
| Specification | Description |
|---|---|
| Number of samples | 24–96 samples (to be processed in increments of 8) |
| Sample input volume | Up to 5 x 105 cells, homogenized in 140 μl Buffer RLT |
| Elution volume | 110 μl |
| Duration | 96 samples in approximately 93 or 114* minutes 24 samples in approximately 54 or 73* minutes |
| Specification | Description |
|---|---|
| Number of samples | 24–96 samples (to be processed in increments of 8) |
| Sample input volume | 350 μl aqueous phase from up to 40 mg frozen or 20 mg stabilized tissue homogenized in 750 μl QIAzol Lysis Reagent |
| Elution volume | 110 μl |
| Duration | 96 samples in approximately 73 or 93* minutes 24 samples in approximately 34 or 54* minutes |
| Specification | Description |
|---|---|
| Number of samples | 24–96 samples (to be processed in increments of 8) |
| Sample input volume | 350 μl aqueous phase from up to 40 mg frozen or 20 mg stabilized tissue homogenized in 750 μl QIAzol Lysis Reagent |
| Elution volume | 110 μl |
| Duration | 96 samples in approximately 78 minutes 24 samples in approximately 38 minutes |
Procedure
With the automated procedure for purification of total RNA from animal or human cells, all RNA molecules longer than 200 nucleotides are purified. This provides enrichment for mRNA, since most RNAs <200 nucleotides (such as 5.8S rRNA, 5S rRNA, and tRNA, which together comprise 15–20% of total RNA) are selectively excluded. The size distribution of the purified RNA is comparable to that obtained by centrifugation through a CsCl cushion, where small RNA does not sediment efficiently.
For purification of total RNA from tissue, or of total RNA including small RNA, from cells and tissues, we recommend first using QIAzol Lysis Reagent as the lysis buffer. Detailed procedures are provided in the kit handbook.
Applications
The RNeasy 96 QIAcube HT Kit provides efficient, high-throughput RNA sample preparation for research use in fields such as drug screening and basic research. The purified RNA is ready to use in any downstream application, including:
- RT-PCR and qRT-PCR
- Differential display
- cDNA synthesis
- Northern, dot, and slot blot analyses
- Primer extension
- Poly A+ RNA selection
- RNase/S1 nuclease protection
Supporting data and figures
Automated purification of RNA from tissues.