qBiomarker Somatic Mutation PCR Assays
For detecting the presence of specific DNA sequence mutations in cancer and oncogenesis
For detecting the presence of specific DNA sequence mutations in cancer and oncogenesis
Cat. No. / ID: 337011
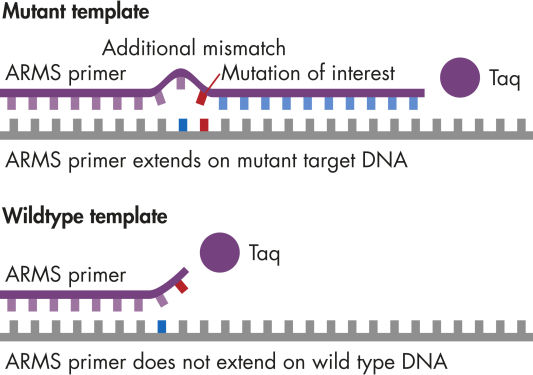
qBiomarker Somatic Mutation PCR Assays identify the presence of individual specific sequence mutations present in cell lines or research samples that are critical for toxicological, drug development, and cancer studies. The mutations are selected from comprehensive curated databases (e.g., COSMIC) and literature reviews based on their clinical or functional relevance and frequency in patient populations.
After the isolation of genomic DNA from fresh, frozen, or fixed samples, add an aliquot of each sample to a separate real-time PCR tube containing the appropriate master mix (included) and the qBiomarker Somatic Mutation PCR Assay. Then, add an aliquot of each sample to a separate real-time PCR tube containing the appropriate master mix (included) and the corresponding reference gene copy assay. Finally, run the recommended cycling program.
Determine the CT values for each mutation-specific assay and the corresponding reference gene copy assay for each sample using your instrument's software. Then, paste the values into the correct Excel-based data analysis template to determine which samples contain the tested mutation.
qBiomarker Somatic Mutation PCR Assays are highly suited for the rapid and accurate identification of individual specific sequence mutations present in fresh, frozen, or fixed samples.