QIAGEN Plasmid Kits for Plasmid DNA Extraction
最大10 mgのトランスフェクショングレードのプラスミドまたはコスミドDNAの精製用
最大10 mgのトランスフェクショングレードのプラスミドまたはコスミドDNAの精製用
✓ オンライン注文による24時間年中無休の自動処理システム
✓ 知識豊富で専門的な製品&テクニカルサポート
✓ 迅速で信頼性の高い(再)注文
カタログ番号 / ID. 12123
✓ オンライン注文による24時間年中無休の自動処理システム
✓ 知識豊富で専門的な製品&テクニカルサポート
✓ 迅速で信頼性の高い(再)注文
QIAGEN Plasmid Kitsは、トランスフェクショングレードのプラスミドDNA精製のための、自然落下式の陰イオン交換チップを備えています。溶解物の不純物除去とイソプロパノール沈殿は遠心分離により行われます。
QIAGEN Plasmid Mega Kit(カタログ番号12181)と QIAGEN Plasmid Giga Kit(カタログ番号12191)は、QIAfilter Mega-Giga Cartridges(カタログ番号19781)と併用することで、遠心分離に代わり、濾過によるバクテリア溶解物に対する急速不純物除去のオプションのプロトコールステップとして使用できます。原理について詳しくは下をご覧ください。
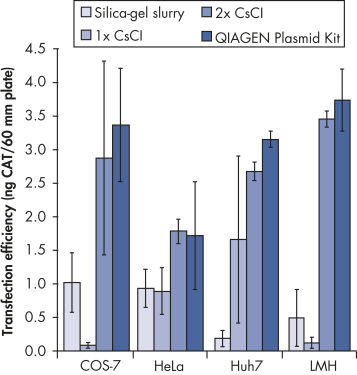
QIAGEN Plasmid Kitsは、自然落下式のQIAGEN陰イオン交換チップを使用して、プラスミドDNAの効率的精製を実現します。最大10 mg(Giga)、2.5 mg(Mega)、500 µg(Maxi)、100 µg(Midi)、20 µg(Mini)の高コピープラスミドDNAを培養液から精製します(培養液の量はプラスミドのコピー数、添加サイズ、宿主株、培養媒体に依存します)。QIAGEN Plasmid Kitsで精製したプラスミドDNAは、トランスフェクション(図「 プラスミド精製法とトランスフェクション効率の比較」を参照)、クローニング、in vitro転写などのアプリケーションでの使用に最適です。
QIAGEN-tips内にある独自の陰イオン交換樹脂は、核酸の精製専用に開発されています。本製品の優れた核酸分離能力により、CsCl密度勾配遠心法を2回連続で行なって得られるDNA純度に匹敵するかそれ以上となります。充填済みのQIAGEN-tipsは、自然落下で作動し乾燥することがなく、プラスミド調製に要する直接の作業時間を短縮できます。QIAGENプラスミド精製システムは全体として、フェノール、クロロフォルム、臭化エチジウム、塩化セシウムなどの有害物質を一切使用していないため、ユーザーにも環境にも影響が最小限になります。
遠心分離の代わりに、濾過による細菌溶解物に対する急速不純物除去のオプションのプロトコールステップとして、QIAfilter Mega-Giga Cartridges(カタログ番号19781)を使用できます。これは掃除機で動作するもので、少ない労力で大量の細菌溶解物でも効率的に不純物除去が行えます。このプロトコールはハンドブックでご覧になれます。
特徴 | Plasmid Mini Kit | Plasmid Midi Kit | Plasmid Maxi Kit | Plasmid Mega Kit | Plasmid Giga Kit |
|---|---|---|---|---|---|
| アプリケーション | トランスフェクション、クローニング、シークエンシング、キャピラリシークエンシングなど | トランスフェクション、クローニング、シークエンシング、キャピラリシークエンシングなど | トランスフェクション、クローニング、シークエンシング、キャピラリシークエンシングなど | トランスフェクション、クローニング、シークエンシング、キャピラリシークエンシングなど | トランスフェクション、クローニング、シークエンシング、キャピラリシークエンシングなど |
| 培養液量/出発物質 | 培養液量3~10 ml | 培養液量25~100 ml | 培養液量100~500 ml | 培養液量500 ml~2.5リットル | 培養液量2.5~5リットル |
| 溶出量 | 可変 | 可変 | 可変 | 可変 | 可変 |
| プラスミドタイプ | 高コピー、低コピー、コスミドDNA | 高コピー、低コピー、コスミドDNA | 高コピー、低コピー、コスミドDNA | 高コピー、低コピー、コスミドDNA | 高コピー、低コピー、コスミドDNA |
| 処理 | 手動(自然落下) | 手動(自然落下) | 手動(自然落下) | 手動(自然落下) | 手動(自然落下) |
| ランあたりのサンプル | ランあたり1サンプル | ランあたり1サンプル | ランあたり1サンプル | ランあたり1サンプル | ランあたり1サンプル |
| 技術 | 陰イオン交換テクノロジー | 陰イオン交換テクノロジー | 陰イオン交換テクノロジー | 陰イオン交換テクノロジー | 陰イオン交換テクノロジー |
| ラン1回あたりの時間 | 80分 | 150分 | 160分 | 220分 | 320分 |
| 収量 | <20 μg | 最大100 µg | <500 µg | <2.5 mg | <10 mg |
QIAGEN Plasmid Kitsを用いて、バクテリア溶解物を遠心分離により不純物除去します。次に、不純物を除去したこの溶解物を陰イオン交換チップに充填し、プラスミドDNAが適切な低塩濃度およびpHの条件下で選択的に結合します。RNA、タンパク質、代謝物、その他の低分子量の不純物は中塩濃度の洗浄で除去します。ゲノムDNA不含の高純度プラスミドDNAを高塩バッファーに溶出します(フローチャート「 QIAGEN Plasmid Kit操作手順」を参照)。このDNAをイソプロパノール沈殿により濃縮、脱塩し、遠心分離で回収します。
QIAGEN Plasmid Kitsで精製されたプラスミドDNAは、以下のようなアプリケーションでの使用に最適です。
| 特徴 | 仕様 |
|---|---|
| Technology | 陰イオン交換テクノロジー |
| Culture volume/starting material | 培養液量3ml~5リットル |
| Yield | <20 µg~<10 mg |
| Processing | 手動(自然落下) |
| Samples per run (throughput) | ランあたり1サンプル |
| Time per run or prep per run | 80~320分 |
| Applications | トランスフェクション、クローニング、シークエンシング、キャピラリシークエンシングなど |
| Plasmid type | 高コピー、低コピー、コスミドDNA |