✓ 24/7 automatic processing of online orders
✓ Knowledgeable and professional Product & Technical Support
✓ Fast and reliable (re)-ordering
Cat. No. / ID: 301105
✓ 24/7 automatic processing of online orders
✓ Knowledgeable and professional Product & Technical Support
✓ Fast and reliable (re)-ordering
Features
- Optimized transfection using cell-specific protocols
- Fast procedure and easy handling
- High cell viability, low cytotoxicity
Product Details
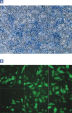
PolyFect Transfection Reagent is based on activated dendrimer technology optimized for the transfection of DNA into COS-7, NIH/3T3, HeLa, 293, and CHO cells.
Performance
PolyFect Reagent consistently delivers high transfection efficiencies (see figures " High transfection efficiencies in CHO and 293 cells" and " PolyFect Reagent with HeLa cells"). In contrast to many liposomal reagents, PolyFect Reagent enables transfection in the presence of serum without lowering of transfection efficiency.
See figures
Principle
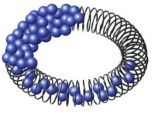
PolyFect Transfection Reagent is a solution of specifically designed activated-dendrimers. The reagent consists of dendrimer molecules of a defined spherical architecture with branches radiating from a central core (see figure " Activated dendrimer"). The branches terminate at charged amino groups, which can interact with negatively charged phosphate groups of nucleic acids. PolyFect Reagent assembles DNA into compact structures (see figure " PolyFect–DNA interaction"), that bind to the cell surface and are taken into the cell by nonspecific endocytosis. The reagent buffers the pH of the endosome, leading to pH inhibitition of endosomal nucleases, which ensures stability of PolyFect–DNA complexes.
See figures
Procedure
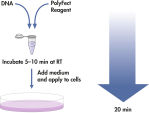
PolyFect Transfection Reagent is provided as a ready-to-use solution. The transfection procedure (see flowchart " PolyFect transfection procedure") is fast and simple — just add PolyFect Reagent to the DNA solution, mix, incubate for 5–10 minutes, add growth medium (which can contain serum and antibiotics), and pipet the PolyFect–DNA complexes onto the cells. The cells are then incubated for gene expression. No post-transfection removal of complexes or medium change/addition is necessary, making the procedure fast and easy.
High-throughput transfection
Transfection using PolyFect Reagent requires minimal handling and removal of transfection complexes is not required, making this reagent highly suitable for high-throughput screening. PolyFect Reagent provides outstanding transfection efficiencies, excellent reproducibility, and minimal cytotoxicity in high-throughput transfection, and is available in bulk quantities. For optimized protocols for transfection of COS-7, NIH/3T3, HeLa, HeLa-S3, 293, and CHO cells in 96-well plates, please contact QIAGEN Technical Services.
See figures
Applications
PolyFect Transfection Reagent is highly suited for efficient transfection of standard cell lines in:
- Studies on gene expression and function
- Drug discovery and development studies
Supporting data and figures
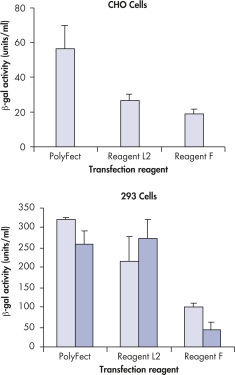
High transfection efficiencies in CHO and 293 cells.
Transfection efficiencies for PolyFect Reagent and 2 lipid-based reagents were compared. Cells cultured in 6-well plates were transfected with a beta-galactosidase-reporter plasmid. The appropriate protocol was used for transfection with PolyFect Reagent. Optimized protocols were used for transfection with Reagents L2 and F; these were: 10 µl Reagent L2 with 3 µg DNA and 15 µl Reagent F with 2 µg DNA for CHO cells, and 10 µl Reagent L2 with 2 µg DNA and 7 µl Reagent F with 3 µg DNA for 293 cells. All transfections were performed in triplicate. Two independent transfection experiments are shown for 293 cells.
Specifications
| Features | Specifications |
|---|---|
| Applications | Plasmid transfection, protein overexpression |
| Transfection type | Transient transfection, stable transfection |
| Controls | Not included |
| Features | Optimized protocols for COS-7, NIH/3T3, HeLa 293, and CHO cells. Transfection in the presence of serum |
| Technology | Activated dendrimers |
| Number of possible transfections | Up to 50–100 transfections in 6-well plates / 1 ml reagent |
| Nucleic acid | DNA |
| Cell type | COS-7, NIH/3T3, HeLa, 293, CHO cells |