QuantiFast Multiplex RT-PCR Kits
For fast, multiplex, one-step qRT-PCR using sequence-specific probes for gene expression analysis
For fast, multiplex, one-step qRT-PCR using sequence-specific probes for gene expression analysis

Cat. No. / ID: 204956

Cat. No. / ID: 204854
QuantiFast Multiplex RT-PCR Kits enable fast and reliable quantification of up to 4 RNA targets in a single tube by multiplex, real-time one-step RT-PCR. A special blend of reverse transcriptases delivers fast and efficient cDNA synthesis. Q-bond technology and an optimized master mix promote fast, multiplex real-time RT-PCR, not only on fast cyclers with short ramping times, but also on standard cyclers. The combination of a hot start and a unique PCR buffer system in the ready-to-use master mix ensures highly sensitive qPCR on any real-time cycler without the need for optimization. Two kit formats are available: the QuantiFast Multiplex RT-PCR Kit for cyclers that require ROX dye for fluorescence normalization, and the QuantiTect Multiplex RT-PCR +R Kit for all other cyclers. For convenience, the master mix can be stored at 2–8°C.
IMPORTANT NOTE: As announced earlier, the production of the QuantiFast Multiplex RT-PCR Kit (400) and the QuantiFast Multiplex RT-PCR +R Kit (400) has been discontinued since mid-2021. Hence, these products will be available only until stocks last. Visit the product page of the successor kit to view improved features or to request a trial kit.
The QuantiFast Multiplex RT-PCR +R Kit (2000) will remain available.
For more information and FAQs on this transition, visit: www.qiagen.com/PCRresource.
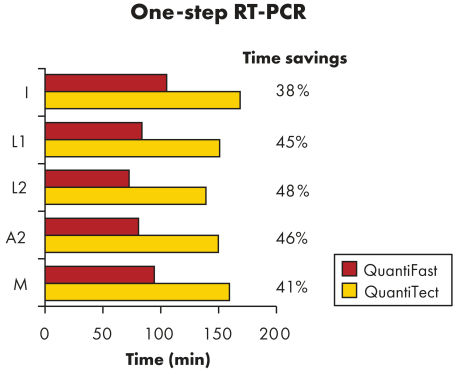
QuantiFast Multiplex RT-PCR Kits reduce RT-PCR run times by up to 50%, allowing you to get results significantly faster (see figure " Significantly reduced RT-PCR times"). You can also greatly increase your sample throughput or efficiently share a cycler with other users. Amplifying control and target genes in the same reaction, instead of in separate reactions, increases the reliability of gene quantification by minimizing handling errors (see figure " Reliable relative quantification"). The special master mix supplied with QuantiFast Multiplex RT-PCR Kits allows rapid setup of multiplex reactions and delivers successful results at the first attempt, providing multiplex RT-PCR data that are comparable with singleplex RT-PCR data (see figure " Comparable results in triplex and singleplex RT-PCR").
QuantiFast Multiplex RT-PCR Kits can clearly distinguish between small differences in the amount of template. Even with two-fold differences in template amount, the kits provide accurate quantification of targets of widely differing abundance. Fast results in multiplex, real-time RT-PCR of up to 4 targets are achieved without compromising performance (see figure " Uncompromised sensitivity in 4-plex RT-PCR").
QuantiFast Multiplex RT-PCR Kits deliver highly sensitive and rapid results over a wide dynamic range on both standard and fast cyclers without optimization (see flowchart " QIAGEN multiplex kits"). The specially developed fast PCR buffer contains the novel additive Q-Bond, which significantly reduces denaturation, annealing, and extension times (see figure " Fast primer annealing").
Amplifying reference and target genes in the same reaction instead of in separate reactions increases the reliability of gene quantification by minimizing handling errors. The QuantiFast Multiplex RT-PCR Buffer includes a balanced combination of K+ and NH4+ ions to promote specific primer annealing, while unique Factor MP stabilizes specifically bound primers (see figure " Unique PCR buffer"). In addition, an optimized mix of reverse transcriptases provides efficient cDNA synthesis in just 20 minutes, while HotStarTaq Plus DNA Polymerase provides a stringent hot start, preventing the formation of nonspecific products.
| Component | Features | Benefits |
|---|---|---|
| HotStarTaq Plus DNA Polymerase | 5 min activation at 95ºC | Set up of qPCR reactions at room temperature |
| QuantiFast Multiplex RT-PCR Buffer | Balanced combination of NH4+ and K+ ions | Specific primer annealing ensures reliable PCR results |
| Synthetic Factor MP | Reliable multiplexing analysis of up to 4 genes in the same tube | |
| Unique Q-Bond additive | Faster PCR run times, enabling faster results and more reactions per day | |
| ROX dye† | Normalizes fluorescent signals on Applied Biosystems and, optionally, Agilent instruments | Precise quantification on cyclers that require ROX dye. Does not interfere with PCR on any real-time cycler |
| QuantiFast RT Mix | Special blend of reverse transcriptases with high affinity for RNA | RNA can be transcribed in just 20 minutes, even through complex secondary structures |
QuantiFast Multiplex RT-PCR Kits contain ready-to-use master mixes that eliminate the need for optimization of reaction and cycling conditions. Simply add template RNA and primer-probe sets to the master mix and follow the protocol in the handbook to get fast and reliable results on any real-time cycler. Kits are available with or without ROX passive reference dye in the master mix, enabling use on virtually any real-time cycler (see table). Due to the optimized ROX concentrations, detection of even low copy numbers is achieved through automatic data analysis.
| ROX dye | Kit | Compatible cyclers |
|---|---|---|
| Supplied in master mix | QuantiFast Multiplex RT-PCR Kit | All cyclers from Applied Biosystems except Applied Biosystems 7500 |
| Supplied in separate tube | QuantiFast Multiplex RT-PCR +R Kit | Applied Biosystems 7500 and cyclers from Bio-Rad, Cepheid, Eppendorf, QIAGEN, Roche, Agilent, and other suppliers |
QuantiFast Multiplex RT-PCR Kits can be used for multiplex gene expression analysis of RNA targets on any real-time cycler. This includes instruments from Applied Biosystems, Bio-Rad, Cepheid, Eppendorf, Roche, and Agilent. For the Rotor-Gene Q and other Rotor-Gene cyclers, we recommend using the Rotor-Gene Multiplex RT-PCR Kit, which has been specially developed for fast cycling on these instruments.
| Features | Specifications |
|---|---|
| Applications | Real-time quantification of RNA targets in a multiplex format |
| Reaction type | Real-time one-step RT-PCR |
| Single or multiplex | Multiplex |
| SYBR Green I or sequence-specific probes | Sequence-specific probes |
| Real-time or endpoint | Real-time |
| Thermal cycler | Real-time cyclers dedicated for multiplex PCR (e.g., most Applied Biosystems real-time PCR cyclers, Roche LightCycler 480, and Bio-Rad iCycler iQ) |
| Sample/target type | RNA |
| With or without ROX | Available with ROX in master mix or with ROX as separate vial |