QIAcuity One, 2plex Device
N° de réf. / ID. 911001
Caractéristiques
- Système entièrement intégré
- Format évolutif (instruments à 1, 4 et 8 plaques)
- Capacités de multiplexage avancées (jusqu’à 12-plex)
- Jusqu’à six canaux standard et deux canaux hybrides pour les colorants LSS
- Cadence d’analyse flexible
- Résultats complets en 2 heures environ
Détails produit
Le QIAcuity Digital PCR System est conçu pour produire des résultats précis de quantification multiplex pour la détection des mutations, les variations du nombre de copies (Copy Number Variation, CNV), les études d’expression génique, l’analyse d’édition génique, et bien plus encore. Ce système à base de nanoplaques intègre un flux de travail de dPCR classique de partitionnement, thermocyclage et imagerie dans une plateforme automatisée et autonome, réduisant au minimum le temps d’intervention.
Ce système s’utilise conjointement avec les nanoplaques, réactifs et dosages QIAcuity.
Explorez la démonstration virtuelle pour en savoir plus sur QIAcuity.
Performances
Le QIAcuity Digital PCR System rend la quantification absolue accessible et abordable pour tous les laboratoires. Permettant un fonctionnement autonome, l’automatisation intègre et simplifie tout le flux de travail de PCR numérique de partitionnement, thermocyclage et imagerie dans un seul instrument avec un temps d’intervention minimal. Il est également facile d’adapter vos dosages de qPCR actuels au QIAcuity Digital PCR System. La manipulation des plaques ne requiert aucune modification par rapport à la qPCR. La configuration des dosages est donc rapide et les résultats sont obtenus en 2 heures environ.
Instruments QIAcuity – fonctionnalités et caractéristiques
| Fonctionnalité | QIAcuity One 2plex | QIAcuity One 5plex | QIAcuity Four | QIAcuity Eight |
|---|---|---|---|---|
| Plaques traitées | 1 | 1 | 4 | 8 |
| Canaux de détection | 2 | 81) (6+2 hybride2) | 81) (6+2 hybride2) | 81) (6+2 hybride2) |
| Capacité de multiplexage | 4 | 123)1) | 123)1) | 123)1) |
| Thermocycleur(s) | 1 | 1 | 1 | 2 |
| Délai d’obtention du résultat | Environ 2 h | Environ 2 h | Première plaque en 2 h environ Une plaque supplémentaire toutes les ∼80 min env. | Première plaque en 2 h environ Une plaque supplémentaire toutes les ∼40 min env. |
| Débit (échantillons traités au cours d’une journée) | Jusqu’à 384 (96 puits) Jusqu’à 96 (24 puits) | Jusqu’à 384 Jusqu’à 96 | Jusqu’à 672 (96 puits) Jusqu’à 168 (24 puits) | Jusqu’à 1248 (96 puits) Jusqu’à 312 (24 puits) |
1) Nécessite l’utilisation du QIAcuity High Multiplex Probe PCR Kit en cas de multiplexage > 5-plex.
2) Les canaux hybrides sont utilisés pour les colorants Long Stokes Shift (LSS).
3) La détection de 12 cibles en parallèle peut être obtenue en utilisant le multiplexage d’amplitude dans les six canaux standard. En combinant les canaux hybrides pour les colorants LSS simplex et le multiplexage d’amplitude, le multiplex total possible peut être augmenté à 14. Cependant, cette combinaison n’est pas recommandée en raison de la demande d’optimisation de tous les dosages utilisés dans un mélange réactionnel et de leur compensation de diaphonie correspondante.
Principe
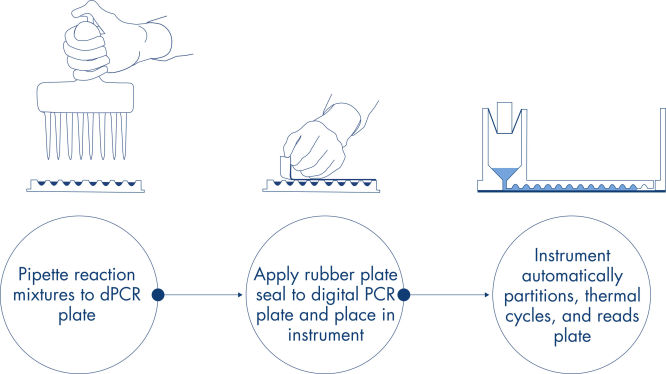
Vous pouvez obtenir, en seulement 3 étapes simples, le résultat de dPCR que vous voulez en 2 heures environ : pipettez et chargez, effectuez l’expérience, puis analysez les résultats.
Vous trouverez ici une description du principe de la réaction de dPCR sur nanoplaques.
Procédure
Comme dans les expériences de qPCR, la préparation des échantillons comprend le transfert du master mix, des sondes et des amorces sur une nanoplaque de 96 ou 24 puits, suivi de l’addition des échantillons. Le système intègre le fractionnement, le thermocyclage et l’imagerie dans un seul instrument entièrement automatisé qui permet aux utilisateurs d’obtenir des résultats en moins de 2 heures. Il est possible d’effectuer l’analyse à l’aide de la suite logicielle, qui donne la concentration de la séquence cible en copies par microlitre et permet le contrôle qualité pour les échantillons positifs ou NTC. Cette analyse peut également être effectuée sur des ordinateurs à distance au sein du même réseau local (Local Area Network, LAN).
Applications
Associés aux QIAcuity Nanoplaques et aux QIAcuity PCR Kits, les instruments QIAcuity facilitent les applications de PCR numérique telles que :
- La détection des mutations rares
- L’analyse des variations du nombre de copies
- L’analyse de l’expression génique
- La détection des agents pathogènes
- Le génotypage
- La recherche sur les miARN
- La thérapie génique et cellulaire
Logiciel
La QIAcuity Software Suite fournie avec l’instrument et installée sur un ordinateur distinct contrôle un ou plusieurs instruments QIAcuity, soit via une connexion directe, soit via un réseau local (Local Area Network, LAN) existant. À l’aide de la QIAcuity Software Suite, les expériences, échantillons et mélanges réactionnels de PCR numérique peuvent être définis, attribués à des nanoplaques et transférés sur l’instrument QIAcuity. Après le cycle d’exécution, il est possible d’analyser les données, de créer des rapports et d’exporter les données pour une analyse externe. Le logiciel offre de multiples fonctionnalités de matrices pour faciliter la réutilisation de plans de plaque ou de paramètres de cycle d’exécution de plaques, transformant encore davantage votre pratique de la PCR numérique.
Lorsqu’il est intégré à un réseau local, l’ordinateur héberge la fonction QIAcuity Software Suite en tant que serveur accessible via LAN aux ordinateurs clients. Cela permet à plusieurs utilisateurs d’accéder au logiciel depuis d’autres pièces ou bureaux et d’analyser les données dans un navigateur classique, sans devoir installer le logiciel sur plusieurs ordinateurs ni utiliser de connexions internet pour accéder aux données et pour les transmettre.
Services
Protégez votre instrument grâce aux diverses solutions de service QIAGEN. Renseignez-vous sur un contrat de service spécifique pour répondre à vos besoins.
Données et illustrations utiles
Une méthode sur plaques simple et rapide
Kits & Consumables
Plans des services
QIAcuity One 2plex, contrat complet
N° de réf. / ID. 9245362
QIAcuity One 2plex, contrat de base
N° de réf. / ID. 9245401
QIAcuity One 5plex, contrat de base
N° de réf. / ID. 9245402
QIAcuity One 5plex, contrat complet
N° de réf. / ID. 9245363
QIAcuity One, Installation & Training
N° de réf. / ID. 9245352
QIAcuity Four, Installation & Training
N° de réf. / ID. 9245353
QIAcuity Eight, Installation & Training
N° de réf. / ID. 9245354