Large-Scale Custom DNA Oligonucleotides
Cat no. / ID. 9224466
Features
- Manufactured to customer specification, supporting custom sequences, formats and order sizes
- Broad range of chemical modifications, dyes and labels for PCR, qPCR and dPCR applications
- High‑purity synthesis suitable for sensitive amplification and repeat use
- Scalable synthesis from routine laboratory use to manufacturing‑scale volumes
- Consistent performance across batches, designed for reliable reordering
- Controlled manufacturing conditions to support contamination-sensitive workflows
Product Details
QIAGEN custom DNA oligos are manufactured to defined customer specifications for workflows that require reliable performance at scale. High-purity primers and probes support PCR, qPCR and dPCR applications with a wide range of sequences, formats and production volumes.
The large-scale oligonucleotide synthesis service is designed for customers who seek repeat supply while maintaining high purity, oligo integrity and batch-to-batch consistency. Multiple delivery formats are available, including liquid, dried and lyophilized oligos, as well as custom labeling and aliquoting options.
Scalable oligo manufacturing enables a smooth transition from development and routine use to manufacturing and production volumes. Consistent processes and reproducible output help reduce repeated optimization efforts when moving to industrial scale applications.
High purity DNA oligos with exceptional lot-to-lot consistency are suitable for sensitive PCR and qPCR assays, including probe based detection. Custom DNA oligos can be manufactured under ISO 9001, ISO 13485 Quality Management System, or according to GMP principles.
Performance
QIAGEN Custom DNA Oligonucleotides are designed for reliable amplification at scale, to deliver consistent performance across batches and order sizes, and to support repeat use in established assays.
High purity synthesis and controlled manufacturing processes help minimize variability, ensuring primers and probes perform predictably as programs transition from routine laboratory use to manufacturing scale production.
Enhanced process controls support contamination sensitive PCR, qPCR and dPCR workflows, making these custom oligonucleotides suitable for long-term, high volume applications where reproducibility and reliability are critical.
| Non-GMP* | GMP† | |
| Quality Management System compliance | ISO 9001 | ISO13485: 2016 and 21CFR part 820 |
| Purification method | Desalted or HPLC | Desalted or HPLC |
| DNA Oligonucleotide length | <170 bp | <170 bp |
| Cross‑contamination control | Standard | Extensive |
| Traceability | Standard | Full |
| HPLC column | Nondedicated | Dedicated |
| Clean room | Nondedicated | Dedicated |
| Product release | Standard final QC | QA review and release |
| Quality documents provided | Certificate of Analysis or Compilation of Results | Certificate of Analysis, including final QC trace ‡ |
| Documentation § | Product batch record | Product batch record with full traceability |
| Environmental control | Standard control | Advanced control |
| Synthesis scale | 40 nmol up to 1000 µmol | 1 – 1000 µmol |
A broad range of chemical modifications, dyes, and labels is supported. Custom DNA synthesis includes commonly used modification technologies such as double quenched probes and BHQ® based quenchers. A detailed view of standard modifications can be found on the Oligo Modifications page.
Principle
Customized DNA oligonucleotides are synthesized using the highly controlled solid-phase phosphoramidite process, where nucleotides are added step by step to a growing chain anchored to a solid support. Each cycle precisely incorporates a specific base, followed by deprotection and purification steps to ensure high sequence accuracy and product quality.
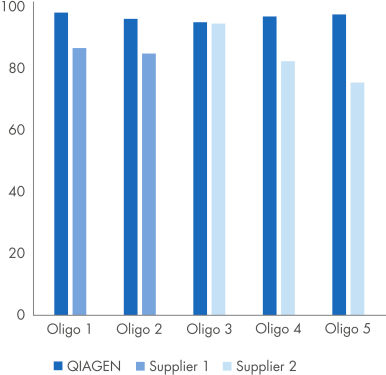
Each primer or probe is synthesized according to defined customer specifications, followed by purification and quality control steps appropriate for PCR and qPCR use, resulting in high purity (see figure “Purity of QIAGEN oligonucleotides compared with purity of oligos from two other suppliers”) and better assay sensitivity (see figure “Comparison in dPCR of RFU (Relative Fluorescence Units) of probes from QIAGEN and two other suppliers”).
Procedure
- Rigorous in-process control and QC analysis guarantee high-quality and full-length sequences
- Precise nucleotide placement in the sequences maximizes specificity, enhancing PCR efficiency
- High purity levels eliminate cross-contamination with other oligonucleotides and reduce off-target binding, ensuring a highly specific and robust PCR reaction
Applications
Custom DNA oligonucleotides are designed to support a wide range of PCR, qPCR and dPCR applications, particularly in workflows that require repeat supply, scalability and consistent performance.
Typical applications include:
- PCR and qPCR assay development and routine use
- Probe-based detection assays, including hydrolysis probe (e.g. TaqMan® type) workflows
- Diagnostics-adjacent and IVD-oriented applications, where quality systems and consistency are important
- High-throughput amplification and screening programs
- OEM and industrial PCR workflows requiring reproducible oligo supply
- Assay scale-up from development to manufacturing use
Services
Dedicated ordering process and project support
Custom DNA oligonucleotides are ordered through a dedicated team that supports specification review, feasibility assessment and quotation for both small- and large-scale requirements.
A dedicated project manager coordinates manufacturing, quality and logistics activities throughout the project lifecycle, supporting smooth progression from development to repeat and large-scale supply.
Direct access to technical experts is available to support assay design considerations, modification selection and troubleshooting related to PCR, qPCR and dPCR applications.
Manufacturing timelines are defined based on oligo specifications, scale and quality level, with a strong focus on reliable, on-time delivery. Once confirmed, delivery dates are consistently maintained, supporting dependable planning for repeat and scale-up orders.
For customers with ongoing demand, oligonucleotides can be manufactured for stock and stored under defined conditions, enabling flexible ordering and release based on actual usage requirements.
Supporting data and figures
Purity of QIAGEN oligonucleotides compared with purity of oligos from two other suppliers
Purity (%) was determined in-house for 5 different oligos (primers and probes) manufactured by QIAGEN and Supplier 1 and Supplier 2. All oligos were claimed to have >90% purity.