✓ 24/7 automatic processing of online orders
✓ Knowledgeable and professional Product & Technical Support
✓ Fast and reliable (re)-ordering
Cat. No. / ID: 53704
✓ 24/7 automatic processing of online orders
✓ Knowledgeable and professional Product & Technical Support
✓ Fast and reliable (re)-ordering
Features
- Rapid purification of high-quality, ready-to-use viral DNA and RNA
- No organic extraction or alcohol precipitation
- Consistent, high yields
- Removal of contaminants and inhibitors
Product Details
The QIAamp UltraSens Virus Kit uses a novel technology to concentrate viral nucleic acids in plasma and serum samples, followed by nucleic acid purification using proven QIAamp technology. Starting with sample volumes of up to 1 ml, nucleic acid concentration is achieved by first adding a novel reagent to the sample. The reagent forms complexes with nucleic acids, allowing them to be highly concentrated by low-speed centrifugation.
Performance
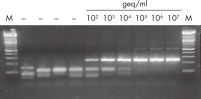
The QIAamp UltraSens Virus Kit is suitable for purification of viral nucleic acids from a wide range of viral species. It can also be used for efficient isolation of extracellular genomic DNA from plasma and serum. The purified nucleic acids perform well in sensitive downstream applications (see figures "High performance in one-step PCR" and " High performance in RT-PCR").
The purified DNA and RNA can be used in a wide range of downstream applications, including:
- PCR and quantitative real-time PCR
- Infectious disease research
See figures
Principle
No phenol–chloroform extraction is required. Nucleic acids bind specifically to the QIAamp silica-gel membrane while contaminants pass through. PCR inhibitors, such as divalent cations and proteins, are completely removed in two efficient wash steps, leaving pure nucleic acids to be eluted in either water or a buffer provided with the kit.
QIAamp technology yields viral RNA and DNA from cell-free body fluids that are ready to use in PCR and blotting procedures. QIAamp sample preparation technology is fully licensed.
Procedure
Nucleic acid concentration is achieved by first adding a novel reagent to the sample. The reagent forms complexes with nucleic acids, allowing them to be highly concentrated by low-speed centrifugation. This step allows nucleic acid purification from large sample volumes without the inconvenience of handling large volumes throughout the protocol. Viral nucleic acids are then purified using QIAamp silica-gel-membrane technology (see flowchart " Procedure"). Lysates are loaded onto the QIAamp spin column. Wash buffers are used to remove impurities and pure, ready-to-use DNA is then eluted in water or low-salt buffer.
No ultracentrifugation or specialized laboratory equipment is required, and an internal control can be added at the start of the procedure, allowing full monitoring of the purification process.
See figures
Applications
The QIAamp UltraSens Virus Kit uses new technology to concentrate viral nucleic acids in plasma and serum samples, followed by nucleic acid purification using proven QIAamp technology. The procedure provides increased sensitivity in viral-load monitoring and other applications where high viral nucleic acid recovery is essential. Samples of 1 ml plasma or serum can be prepared within 1 hour, with typical yields of >85% recovery of nucleic acids and 60 µl elution volume.
Supporting data and figures
QIAamp UltraSens procedure.

Specifications
| Features | Specifications |
|---|---|
| Applications | PCR, qPCR, real-time PCR. |
| Time per run or per prep | 1 hour |
| Format | MinElute columns |
| Elution volume | 60 µl |
| Processing | Manual (centrifugation) |
| Sample amount | 1 ml |
| Technology | Silica technology |
| Purification of total RNA, miRNA, poly A+ mRNA, DNA or protein | Viral DNA, viral RNA |
| Main sample type | Serum, plasma |
| Yield | >85% recovery |