✓ 24/7 automatic processing of online orders
✓ Knowledgeable and professional Product & Technical Support
✓ Fast and reliable (re)-ordering
Cat. No. / ID: 937556
✓ 24/7 automatic processing of online orders
✓ Knowledgeable and professional Product & Technical Support
✓ Fast and reliable (re)-ordering
Features
- For in vitro diagnostic use
- Lot-to-lot traceability
- Produced under GMP manufacturing conditions
Product Details
Principle
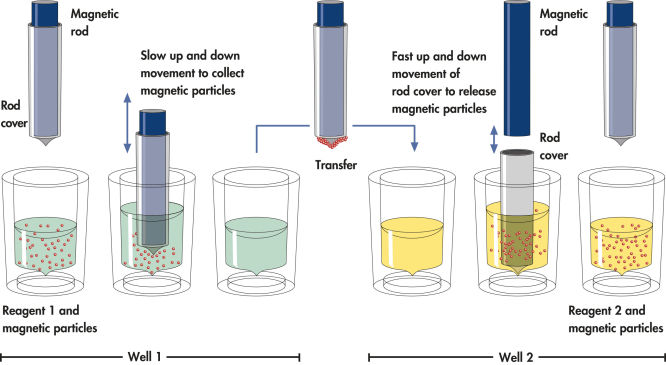
QIAsymphony technology combines the speed and efficiency of anion exchange-based nucleic acid purification with the convenient handling of magnetic particles (see figure “ Schematic diagram of the QIAsymphony SP principle.”).
The QIAsymphony SP processes a sample containing magnetic particles as follows: A magnetic rod protected by a rod cover enters a well containing the sample and attracts the magnetic particles. The magnetic rod cover is positioned above another well and the magnetic particles are released. These steps are repeated several times during sample processing The QIAsymphony SP uses a magnetic head containing an array of 24 magnetic rods, and can therefore process up to 24 samples simultaneously.
See figures
Procedure
The QIAsymphony SP makes automated sample preparation easy and convenient. Samples, reagents and consumables, and eluates are separated in different drawers. Simply load samples, proteinase K, reagents provided in special cartridges, and preracked consumables in the appropriate drawer before a run. Start the protocol and remove purified DNA from the “Eluate” drawer after processing. Refer to the user manuals supplied with your instrument for operating instructions.
The purification procedure is designed to ensure safe and reproducible handling of potentially infectious samples, and comprises 3 steps: bind, wash, and elute (see figure “ QIAsymphony DSP Circulating DNA procedure.”). The user can choose between different sample input volumes.
See figures
Applications
ccfDNA purified using the QIAsymphony DSP Circulating DNA Kit and the QIAsymphony SP is ready for use in a wide range of downstream applications, including NGS, digital, multiplex and quantitative real-time PCR.
Supporting data and figures
Efficient processing of magnetic particles.